There is a page containing 8 multiple choice questions and the project rubric. Worksheets are Atomic timeline, History of the atom timeline answers, Atomic theory timeline, Chapter 2 reinforcement work atomic timeline denton isd, Basic atomic theory the structure of matter, Work development of atomic theory name true false, Activity 1. Some students get creative by decorating with ways to remember the models: "chocolate chip cookie dough" around Thomson's information or "Legos" around Democritus'. Displaying all worksheets related to - Atomic Theory Timeline. I encourage them to make the poster colorful in order to separate the information that belongs together. The students read through the Information Book, cut out the "Everything You Need" pages and organize the images and information boxes into their own timeline. The Information Book is now also included as a digital Google document so you can share it with your students on Google Classroom and save paper! They end up being the perfect size to fit all of the information/pictures, and they look great hung up in my classroom and are not too big!Ĭopy one "Information Book" (it looks fine in Black and White - I copy it back-to-back, put the pages into four plastic sleeves, and bulldog clamp the pages for use each year) and one set of the Directions and "Everything You Need to Make an Atomic Theory Timeline" per group or pair of partners. 
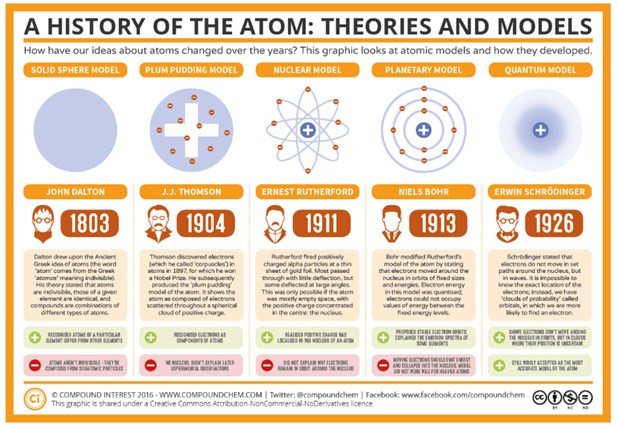
I provide them with 11" by 17" white construction paper for the "posters". I like to show this Ted Ed video before we begin: This lesson plan covers major developments and changes in atomic theory, with a focus on the 1800s and 1900s. This project covers: Democritus, Aristotle and "the Death of Chemistry", Dalton, Thomson, Rutherford, Bohr, and the Modern Cloud Theory. Nuclear fission is what creates energy, through the separation of uranium atoms.The Atomic Theory Timeline Project is everything you need to give your chemistry students a nice background on the history of the atom! Students use skills of synthesizing informational text, organizing pieces of information into groups and then into a chronological order, and displaying the information in a clear and creative way. The modern-day application of chemists quest to refine and purify substances is demonstrated at a solar panel plant where a common material silica sand is transformed into. Otto Hahn and Lise Meitner were the first to discover nuclear fission. The contributions to atomic theory of Dalton, Proust, Lavoisier, as well as those of the Arabic scientist, Jbir ibn ayyn, who died in 803 AD, are discussed. In this study guide, you can revise how the periodic table arranges elements according to. 1934:ġ1 Otto Hahn and Lise Meitner were the first to discover nuclear fission Atoms are made from protons, neutrons and electrons. Thomsons greatest contribution to science to be his role as a teacher. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. His atomic theory helped explain atomic bonding and the structure of molecules. This means that it is very well possible to change the atoms in matter. Thomson was closely aligned with chemists of the time. 1926:ġ0 Irene Curie and Frederic Joliot-Curie discovered that it’s possible to create artificial radioactive elements. This equation is required for quantum mechanics. The Shrodinger Equation is named after him. 1909:ĩ Erwin Shrodinger discovered that electrons move in the form of a waveĮrwin Shrodinger discovered that electrons move in the form of a wave. He was the first one to discover the atomic nucleus. 1908:Ĩ Ernest Rutherford has made a theory on the structure of atoms, what they look like. He did this through an experiment, the ‘Oil Drop Experiment’. Some scientists will have more than one description associated with them. Your task today is to match the scientist with the appropriate description. Robert Millikan was the first to measure an electron for its charge. Chapter 14 - Atomic Theory Scientist Timeline Below is a list of individuals that have made significant contributions to the concept of the atomic theory. This makes heat, which makes steam, which is used to make electricity.ħ Robert Millikan was the first to measure an electron for its charge This is done by fission (when uranium atoms split). The main use of nuclear energy is to produce electricity. 1896:ĥ JJ Thomson was the first to discover electrons.Ħ 1905: Albert Einstein made a hypothesis about the particle of light. 442 BC:ģ John Dalton discovered The Atomic Theory.Ĥ Wilhelm Rontgen discovered that some chemicals would glow under cathode rays. This was when the later to be known as ‘The Atomic Theory’ started. 
He believed that it was impossible to destroy atoms, and that there were many different kinds of atoms. He was also known to be the ‘father of modern science’. 2 Democritus was the first to think that all matter was made of indivisible elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
